Tackling the Top 5 Stability Challenges in Drug Development
In drug development, stability isn’t just a checkpoint, it’s the foundation of whether a therapy makes it to patients. A single instability issue can set a program back months or even years.
We’ve gathered the five of the most common risks and how scientists are addressing them.

5 Factors That Impact Drug Stability

1. Solubility and Bioavailability
What is the Risk?
Poor solubility and low permeability in active pharmaceutical ingredients (APIs), especially in BCS Class II and IV drugs, can hinder the body’s ability to absorb the drug properly.
Why it Matters?
Solubility is essential for drug success. If left unaddressed it can cause a delay in development and potential failure in preclinical or clinical stages.
Innovative Strategies at Work
Implementing advanced formulation strategies including solid dispersion, amorphous transformation and active polymorph screening help identify the most stable and bioavailable forms earlier in development. In the early stages of drug development, predictive solubility modeling helps researchers understand how a molecule behaves under specific conditions, leading to better earlier formulation decisions.
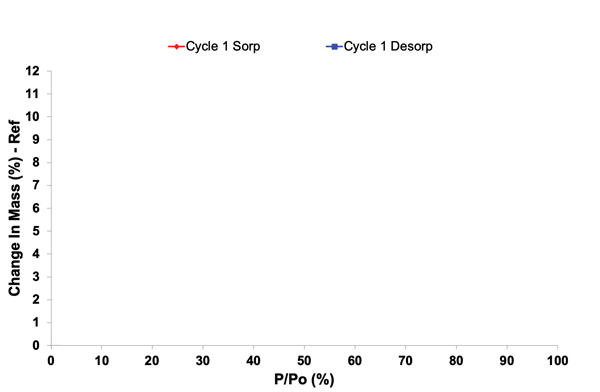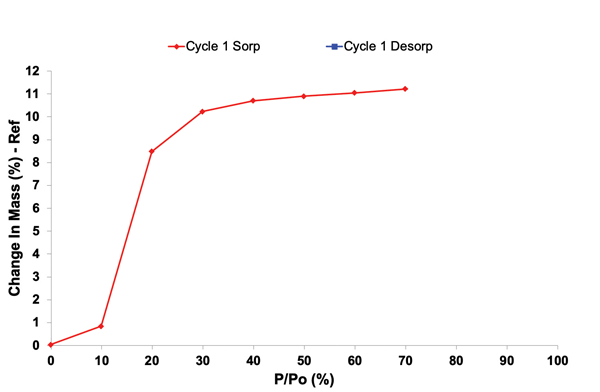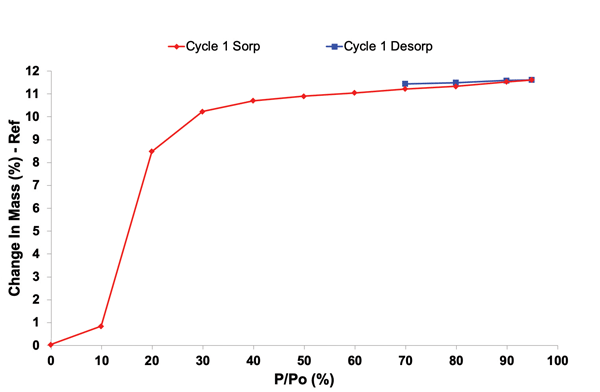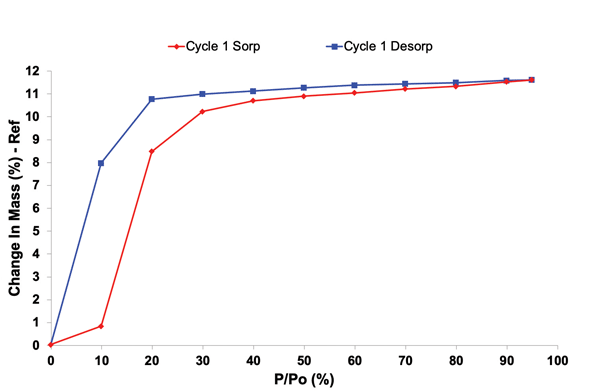
2. Environmental Conditions
What is the Risk?
Moisture, heat, and oxygen can degrade API and excipients causing chemical changes, like hydrolysis, due to moisture absorption from the environment. They can also cause polymorphic transitions can occur during storing and processing. Predicting shelf life is difficult without thorough stability analysis.
Why it Matters?
Environmental destabilization is one of the leading causes of formulation failure affecting the product’s safety, potency, and regulatory compliance which could lead to recalls or reformulation.
Innovative Strategies at Work
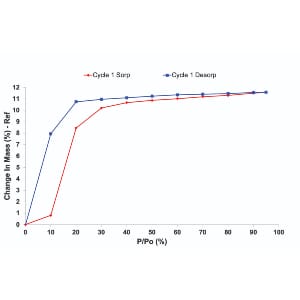
Stability profiling under controlled temperature and humidity conditions helps predict shelf life, guide packaging decisions, and design formulations that withstand real-world conditions. Advanced solid-state tools that provide insights into stability profiling, include Dynamic Vapor Sorption and Inverse Gas Chromatography (iGC). They can provide insights into how materials respond including adsorption kinetics, surface energy, and surface morphology.

3. Drug-Excipient Interactions
What is the Risk?
Excipients are often considered “inactive” ingredients, but their interactions with APIs could affect a drug’s stability by modifying its release profile or therapeutic effectiveness. Choosing an excipient that enhances solubility and without causing degradation can be difficult in early development stages.
Why it Matters?
Incompatible excipients can sabotage otherwise strong formulations wasting time, money, and also increases chances of formulation failure in late stages.
Innovative Strategies at Work
Compatibility studies, using tools like calorimetry or spectroscopy is standard and techniques like Inverse Gas Chromatography (iGC) help detect hidden incompatibilities early. Using the iGC to understand moisture affinity, surface chemistry, and reactivity, helps formulators select excipients that enhance performance without compromising stability.

4. Process and Scale-up Challenges
What is the Risk?
Success in lab research doesn’t always translate to successful full scale manufacturing. An increase in production volume can cause changes in granulation, drying temperature, and mixing strength. These issues can lead to powder flow issues, poor compressibility, and batch-to-batch inconsistency.
Why it Matters?
Scale-up challenges can result in production halts, corrupted test batches and regulatory delays if processes don’t result in consistent and reproducible results.
Innovative Strategies at Work
Quality by Design (QbD) and Process Analytical Technology (PAT) approaches help define critical material attributes and process parameters. Powder flow testing and material characterization ensure scalability before full-scale production. These insights allow teams to anticipate processing behavior, adjust conditions early, and avoid costly last-minute rework during scale-up.

5.Moisture and Sorption Behavior
What is the Risk?
APIs and excipients are moisture sensitive, which can lead to crystallization, amorphization, or chemical degradation. Precise models of sorption behavior is essential to accurately predict how these changes will impact a drug’s stability, performance, and shelf life.
Why it Matters?
Uncontrolled moisture uptake can reduce product shelf-life resulting in either over-engineering or under-protecting the drug.
Innovative Strategies at Work
Humidity-controlled studies and sorption profiling reveal how APIs and excipients interact with water vapor. When combined with techniques like iGC, formulators gain a complete picture of moisture behavior, packaging needs, and long-term stability.

The Role of Regulatory Guidelines in Drug Stability
Across all these risks, stability testing is guided by international standards from the FDA, EMA, NMPA, and Pharmacopeias (USP, Ph. Eur., ChP). Compliance ensures stability data is valid, comparable, and accepted globally, paving the way for faster approvals and safer launches.

What can you do?
Stability challenges are a constant in pharmaceutical development, but with the right strategies, they don’t have to become roadblocks.
By combining predictive testing, advanced analytical tools, and smart formulation design, scientists can anticipate risks early, protect their compounds, and ultimately bring safer, more effective therapies to patients faster.
Check out our case study on pharmaceutical solid-state stability using DVS and IGC-SEA to analyze polymorphs, amorphous content, moisture sorption, and glass transition for more insights.