Unlocking Breakthroughs:
8 Ways DVS is Essential to
Pharmaceutical Research
Moisture sorption properties stand at the forefront of pharmaceutical material considerations, exerting profound effects on storage, stability, processing, and performance. Guided by the stringent standards of regulators around the world, moisture, far from being a mere impurity, commands strict monitoring and control, especially within drug substances.
The implications of moisture content extend across critical facets of pharmaceutical products, influencing crystallinity, storage modulus, permeability, density, and the melting point, with a pronounced impact on amorphous materials. Recognizing the need for a nuanced approach to moisture analysis, the Dynamic Vapor Sorption (DVS) technique was developed by Surface Measurement Systems in the early 1990s.
This technique, now a cornerstone in pharmaceutical research, offers a rapid, highly-sensitive, and automated method for studying moisture sorption properties across a spectrum of materials. Below we unravel the multifaceted benefits of employing DVS in the analysis of pharmaceutical materials.


1. Compliance with pharmaceutical regulations
The Dynamic Vapour Sorption (DVS) technique is instrumental in meeting stringent US pharmaceutical standards by providing a rapid, highly sensitive, and continuous approach to studying moisture sorption properties. Developed by Surface Measurement Systems, the DVS instrument ensures compliance with US Pharmacopeia guidelines by actively measuring and controlling relative humidity and a wide range of organic vapour concentrations. This precision in environmental control enhances understanding across various applications, including the determination of moisture content, sorption isotherms, and the characterization of hydrates and solvates. Additionally, DVS proves valuable in assessing hygroscopicity, a crucial factor in preformulation activities and the selection of drug crystal forms.

2. Provides unparalleled insight into moisture-induced phase changes
Understanding moisture-induced phase changes is vital in the development of pharmaceutical materials, with a focus on crystallinity, amorphous substances, and hydrate and solvate formation. DVS enables precise examination of amorphous solids, crucial for understanding the critical humidity at which glass transitions occur, impacting storage and processing conditions. Linear relative humidity ramping experiments and 2-dimensional phase diagrams contribute to determining optimal stability conditions for amorphous pharmaceutical ingredients.
Furthermore, DVS plays a vital role in characterizing hydrate and solvate formation, offering valuable data for pharmaceutical development. It detects different hydrate forms and studies stoichiometric solvates, providing a versatile methodology for various solvent concentrations. The integration of DVS with in-situ vibrational spectroscopy enhances the understanding of moisture-induced phase changes by monitoring molecular vibrational characteristics. In summary, DVS is an indispensable tool for unravelling the complexities of moisture-induced phase changes in pharmaceuticals, encompassing crystallinity, amorphous materials, and hydrate and solvate formation.
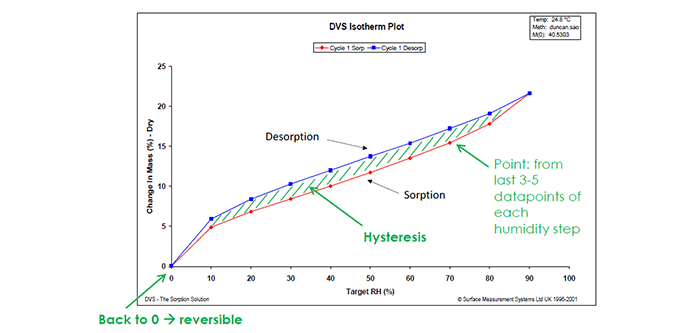
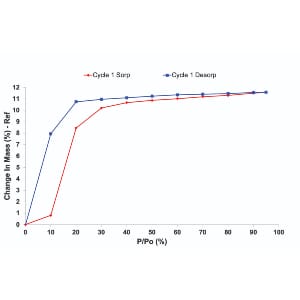
3. Understand the moisture sorption hysteresis of your materials
When attempting to analyse moisture sorption hysteresis, the DVS technique has a unique ability to swiftly measure sorption kinetics and determine moisture diffusion coefficients, facilitating a comprehensive understanding of material transformations. This rapid and continuous measurement capability is in stark contrast to the prolonged timelines associated with traditional desiccator jar methods. DVS’s role extends beyond the laboratory, offering insights into formulation optimization, manufacturing, and quality control processes. By studying moisture sorption hysteresis, researchers can make informed decisions about storage requirements, ensuring the stability and performance of pharmaceutical materials. The instrument’s simultaneous determination of sorption and desorption isotherms on the same sample is particularly valuable, shedding light on dynamic material behaviour under varying humidity conditions. This nuanced understanding aids in tailoring formulations for optimal performance, ultimately contributing to the efficiency and quality of pharmaceutical production.

4. More than just powders – gain vital insight on pharma packaging materials
Dynamic Vapour Sorption (DVS) emerges as an indispensable tool in pharmaceutical research, particularly in the realm of studying packaging materials. DVS facilitates the measurement of diffusion coefficients for films, powders, and fibers, providing valuable insights into the permeability and transport properties of pharmaceutical packaging materials. By conducting real-time mass change experiments, DVS allows for the determination of diffusion coefficients over a range of temperatures, offering a nuanced understanding of how these materials interact with water vapor. This capability is particularly beneficial for packaging applications, where the diffusion into films can be critical for ensuring the stability and integrity of pharmaceutical products during storage and transportation.
Furthermore, DVS proves instrumental in evaluating water vapor transmission rates (WVTR) for pharmaceutical packaging materials. The innovative Payne style diffusion cell, designed for this purpose, enables the measurement of the rate of diffusion of water vapor through thin films. This information is crucial for assessing the effectiveness of packaging barriers in preventing moisture ingress, which is vital for maintaining the quality and shelf-life of pharmaceutical products. The combination of diffusion coefficient determination and WVTR measurement using DVS provides comprehensive data for optimizing pharmaceutical packaging, contributing to the development of packaging solutions that meet stringent quality standards and regulatory requirements.
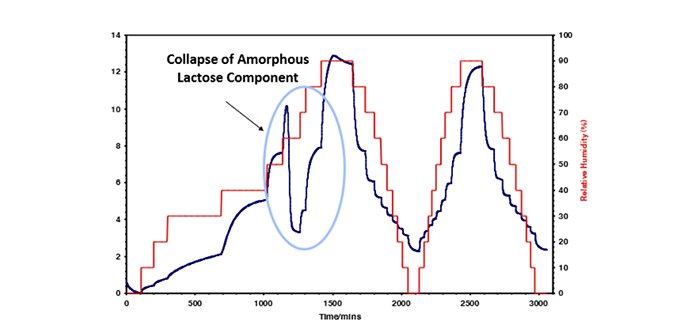
5. Gain detail insight into sorption mechanisms of pharma materials
Having a detailed understanding of the sorption mechanisms of any given material is essential in determining the effective use of pharmaceutical materials, including excipients, drug formulations, and packaging films. One significant benefit of DVS is its ability to generate moisture sorption isotherms, providing crucial insights into the material’s response to varying relative humidity (RH) levels. Unlike traditional methods such as the desiccator jar, DVS enables continuous measurement of sorption kinetics, allowing for the determination of moisture diffusion coefficients and the measurement of sorption and desorption isotherms on the same sample. This real-time capability significantly accelerates the process, reducing the time required for determining isotherms from weeks or months to hours or days. Additionally, DVS facilitates the measurement of moisture-induced phase changes, aiding in the understanding of critical humidity thresholds for glass transitions and potential issues such as powder caking.
As previously mentioned, the DVS technique proves instrumental in characterizing hydrates and solvates of pharmaceutical materials. It enables the detection and characterization of hydrate formation based on environmental relative humidity, allowing for a deeper understanding of the influence of hydration states on physico-chemical properties. DVS can also extend its applicability to studying stoichiometric solvates, providing valuable information on solvate formation and stability. The technique’s combination with in-situ vibrational spectroscopy enhances its capabilities, allowing for the monitoring of molecular vibrational characteristics during hydration state changes. Overall, DVS emerges as a powerful tool for predicting dispersion in liquid solvents, offering insights into sorption kinetics, isotherm characteristics, and the formation of hydrates and solvates critical for pharmaceutical development and quality control.
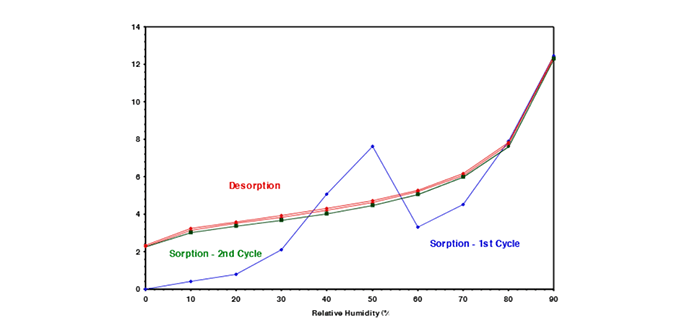
6. Understand the importance of sorption and desorption curves with continuous measurement of sorption kinetics
Crucial insights into moisture sorption properties through the analysis of sorption and desorption isotherms are vital to ensuring stability in materials for new coatings and treatments. By continuously measuring sorption kinetics in real-time, DVS enables the determination of moisture diffusion coefficients and the exploration of moisture sorption hysteresis on the same sample. This dynamic approach accelerates data collection, offering a nuanced understanding of how pharmaceutical materials interact with water vapor at various relative humidity levels, essential for assessing storage conditions and preventing potential degradation risks.
Sorption kinetics data obtained from DVS play a pivotal role in developing coatings and treatments aimed at maintaining product stability. Understanding the rate of moisture absorption and release empowers researchers to design precise coatings, preventing moisture-induced phase changes that could compromise the stability of amorphous pharmaceutical ingredients. DVS’s real-time, detailed sorption kinetics data facilitate the customization of coatings and treatments, ensuring materials withstand environmental humidity, contributing to the formulation of pharmaceutical products that meet stringent stability and quality requirements throughout their lifecycle.

7. Impact of moisture in Hydrolysis and Drug Degradation
Crucial insights into moisture sorption properties through the analysis of sorption and desorption isotherms are vital to ensuring stability in materials for new coatings and treatments. By continuously measuring sorption kinetics in real-time, DVS enables the determination of moisture diffusion coefficients and the exploration of moisture sorption hysteresis on the same sample. This dynamic approach accelerates data collection, offering a nuanced understanding of how pharmaceutical materials interact with water vapor at various relative humidity levels, essential for assessing storage conditions and preventing potential degradation risks.
Sorption kinetics data obtained from DVS play a pivotal role in developing coatings and treatments aimed at maintaining product stability. Understanding the rate of moisture absorption and release empowers researchers to design precise coatings, preventing moisture-induced phase changes that could compromise the stability of amorphous pharmaceutical ingredients. DVS’s real-time, detailed sorption kinetics data facilitate the customization of coatings and treatments, ensuring materials withstand environmental humidity, contributing to the formulation of pharmaceutical products that meet stringent stability and quality requirements throughout their lifecycle.

8. Unlock advanced applications with organic solvents sorption analysis
When applied with organic solvents, the Dynamic Vapour Sorption (DVS) technique unlocks advanced applications that significantly contribute to characterizing and determining physicochemical properties, particularly surface area, in pharmaceutical materials. DVS instruments, with their unique capability to actively measure and control the concentration of a broad range of organic vapours, extends its utility beyond water vapour analysis. This innovative feature allows researchers to explore the interaction between pharmaceutical materials and organic solvents in real time. Notably, the DVS technique applied to organic solvents proves instrumental in determining surface area using the BET (Brunauer-Emmett-Teller) method. Unlike traditional volumetric techniques, DVS experiments are performed at atmospheric pressure and room temperature, mitigating the risk of altering the fragile structure of materials.
The application of DVS with organic solvents is exemplified in the determination of the BET surface area for pharmaceutical materials, such as Metformin Hydrochloride, showcasing the technique’s advantages over conventional methods. The dynamic flow nature of DVS enables rapid equilibration, providing efficient and accurate measurements of surface area with relatively small sample sizes. This capability is particularly advantageous for new drug entities or materials with very low surface areas. Therefore, the DVS technique, when coupled with organic solvents, emerges as a powerful tool for pharmaceutical researchers, offering a dynamic and precise approach to surface area determination and advancing the characterization of materials critical for drug development and quality control.

Conclusion:
In conclusion, the Dynamic Vapour Sorption (DVS) technique presented here demonstrates its pivotal role in unraveling the complexities of moisture sorption, hydrate/solvate formation, and physicochemical properties in pharmaceutical materials. Its wide range of applications, from understanding moisture-induced phase changes to characterizing surface area with organic solvents, showcases the versatility and precision of DVS technology.
For those seeking to delve deeper into these advanced applications and explore the full potential of our DVS, find out more about our products on our company page, or read a full Pharma Overview Scientific Note for DVS using the link below.


Season’s Greetings from Surface Measurement Systems
| It’s been an important year for Surface Measurement Systems globally, full of bold new steps for the business. Among the many exciting developments was the launch of our new contract analysis business, Particle Characterization Laboratories, in Hyderabad, India back in February. From there we’ve continued to grow and expand our sales and service teams across the world, elevating our customer engagement even further. This is coupled with an expansion of our research and development as well as the science teams, to support our plans for product development. A key feature of 2023 has been new faces! As we expand the global ambitions for the business, I have had the pleasure of welcoming many new staff to Surface Measurement Systems globally.
The last 12 months saw the release not only of a brand-new website and online customer portal (www.sorptionhub.com), but Surface Measurement Systems also launched ground-breaking new instruments for developing new carbon capture materials; the DVS Carbon family. These products are being closely followed by two innovative Break Through Analysis (BTA) products; the BTA Horizon and BTA-Frontier. And of course, we continue to improve and develop our current products, including a new family of improved microbalances and an updated IGC-SEA product with a high temperature capability. We look forward to continuing to serve the global sorption community with innovative solutions, unparalleled aftersales care, world-leading scientific support in an exciting 2024. Festive greetings from the Surface Measurement Systems team! |

Prof. Daryl Williams, Founder & MD of SMS, Inducted as a Fellow of the Royal Academy of Engineers
DISCLAIMER: Story not to be shared on social media
Among the many exciting developments at Surface Measurement Systems in 2023, our Founder & MD, Prof. Daryl Williams, was recognized for his lifelong contributions to the field of engineering by being elected a Fellow of the Royal Academy of Engineering.
Along with 73 other leading figures in the field of engineering and technology, Prof. Williams was inducted into the Academy in a special ceremony on 28th November, officiated by HRH Princess Anne. In joining the Fellowship, they will lend their unique capabilities to achieving the Academy’s overarching strategic goal to harness the power of engineering to create a sustainable society and an inclusive economy for all.


More about Prof. Daryl Williams
Professor Daryl Williams is an internationally recognised expert in particle engineering and inventor of novel powder surface characterisation instruments, globally commercialised by his Surface Measurement Systems. The Dynamic Vapour Sorption (DVS) technique allows rapid determination of moisture sensitivity of powders, particularly drug formulations. It is used by every major pharmaceutical company in the world and in more than 1,000 universities and research laboratories.
His research group at Imperial College has consistently produced solutions for international companies on materials manufacture, characterization, and performance. As academic lead for the Imperial College Carbon Capture Pilot Plant he has pioneered hands-on engineering teaching, outreach and research on climate change mitigation.

A Message from Daryl
| It’s been an important year for Surface Measurement Systems globally, full of bold new steps for the business. Among the many exciting developments was the launch of our new contract analysis business, Particle Characterization Laboratories, in Hyderabad, India back in February. From there we’ve continued to grow and expand our sales and service teams across the world, elevating our customer engagement even further. This is coupled with an expansion of our research and development as well as the Science teams, to support our plans for product development. A key feature of 2023 has been new faces! As we expand the global ambitions for the business, I have had the pleasure of welcoming many new staff to Surface Measurement Systems globally.
The last 12 months saw the release not only of a brand-new website and online customer portal (www.sorptionhub.com), but Surface Measurement Systems also launched ground-breaking new instruments for developing new carbon capture materials; the DVS Carbon family. These products are being closely followed by two innovative Break Through Analysis (BTA) products; the BTA Horizon and BTA-Frontier. All of these products are receiving strong market interest. Of course, we continue to improve and develop our current products including a new family of improved microbalances and an updated IGC-SEA product including a high temperature capability. I would like to thank all of the Surface Measurement Systems staff for their dedication and hard work performed in 2023- we really do appreciate it. We look forward to continuing to serve the global sorption community with innovative solutions, unparalleled aftersales care, world-leading scientific support in an exciting 2024. Festive greetings from the Surface Measurement Systems team! |

SMS Employee Milestones
A few of our veteran employees have crossed some big milestones in the last year! We wanted to highlight the colleagues who have passed a significant market in the length of service they have dedicated to the company.
Thank you all for your invaluable contributions, we couldn’t have done it without you!


New starters & Qualifications
Important Milestones for the SMS Team
The team at Surface Measurement Systems continues to expand, with every office seeing new members join us for the exciting times ahead! As well as this, are existing team mates are making huge strides in their own development, marking off some big achievements in the past 6 months.
Below is a quick review of who has joined us in the last few months, and who has passed some big milestones in their own personal development.

New Team members
Qualifications

Prof. Daryl Williams, Founder & MD of SMS, Inducted as a Fellow of the Royal Academy of Engineers
DISCLAIMER: Story not to be shared on social media
Among the many exciting developments at Surface Measurement Systems in 2023, our Founder & MD, Prof. Daryl Williams, was recognized for his lifelong contributions to the field of engineering by being elected a Fellow of the Royal Academy of Engineering.
Along with 73 other leading figures in the field of engineering and technology, Prof. Williams was inducted into the Academy in a special ceremony on 28th November, officiated by HRH Princess Anne. In joining the Fellowship, they will lend their unique capabilities to achieving the Academy’s overarching strategic goal to harness the power of engineering to create a sustainable society and an inclusive economy for all.


More about Prof. Daryl Williams
Professor Daryl Williams is an internationally recognised expert in particle engineering and inventor of novel powder surface characterisation instruments, globally commercialised by his Surface Measurement Systems. The Dynamic Vapour Sorption (DVS) technique allows rapid determination of moisture sensitivity of powders, particularly drug formulations. It is used by every major pharmaceutical company in the world and in more than 1,000 universities and research laboratories.
His research group at Imperial College has consistently produced solutions for international companies on materials manufacture, characterization, and performance. As academic lead for the Imperial College Carbon Capture Pilot Plant he has pioneered hands-on engineering teaching, outreach and research on climate change mitigation.

Blog: Evaluating Materials for Carbon Capture, Unveiling the Power of DVS Carbon
The growing need to combat climate change has intensified the quest for sustainable solutions, leading researchers to explore innovative methods for carbon capture, utilization, and storage (CCUS).
Among the wide array of materials under scrutiny, solid sorbents have emerged as promising candidates, offering unique mechanisms for capturing and storing Carbon Dioxide (CO2). In this blog post, we unravel the transformative capabilities of the DVS Carbon, a high-resolution gravimetric sorption analyzer, and its pivotal role in evaluating materials for CCUS applications.

The Landscape of Solid Sorbents in CCUSAs the urgency to decarbonize our society accelerates, the scientific community is navigating a complex landscape of materials that have potential in addressing the challenges of carbon capture. From metal-organic frameworks (MOFs) to zeolites and activated carbons, researchers are exploring diverse avenues to find materials that can efficiently and sustainably capture CO2. The efficacy of these materials, however, hinges not only on their capacity to absorb CO2 but also on their performance under realistic and dynamic process conditions. Challenges in Material EvaluationEffectively evaluating materials for CCUS demands a multifaceted approach. Beyond assessing the quantity of CO2 a material can absorb, researchers must consider a spectrum of factors such as working capacity, kinetics, selectivity, stability, thermal properties, and cost considerations. A recent collaboration between researchers at Imperial College in London and industry experts resulted in the formulation of best practices, emphasizing the need for complete characterization metrics and in-process relevant conditions. |
The Crucial Role of DVS Carbon in Material Evaluation
In this landscape of complex challenges and evolving best practices, the DVS Carbon stands as stands out as a promising solution. Developed with the express purpose of enabling precise and realistic testing of materials for CCUS, the DVS Carbon is a gravimetric sorption analyzer that transcends traditional measurement tools. Let’s explore the key features that make the DVS Carbon an indispensable instrument in the pursuit of sustainable carbon capture solutions.
Gravimetric Precision and Real-Time MonitoringAt the heart of the DVS Carbon lies its gravimetric basis, leveraging a highly sensitive symmetric SMS Ultrabalance. This microbalance facilitates direct measurements of changes in sample mass with unparalleled precision, reaching down to the microgram level. This real-time monitoring capability with sub-second resolution is foundational in understanding the dynamic nature of material interactions with CO2 under varying conditions. Dynamic Flow System for Realistic ConditionsThe DVS Carbon’s dynamic flow system is a cornerstone of its capability to simulate realistic process conditions. By precisely controlling CO2 concentration, flow rates, temperature and humidity, researchers can mimic diverse scenarios, from Direct Air Capture with low CO2 concentrations to post-combustion conditions with high temperature and CO2 gas concentrations. This flexibility is instrumental in obtaining insights into material performance that align with real-world applications. Innovation in Sensor TechnologyTo enhance its performance, the DVS Carbon incorporates patented speed-of-sound sensors. This sensor technology, specially redesigned for this purpose, enables the high-accuracy measurement of carbon dioxide concentrations in the gas phase. Operating within a range of up to 100 percent CO2 by volume, these sensors contribute to the instrument’s ability to capture nuanced changes during absorption and desorption processes. In the following sections, we will delve into case studies that exemplify the DVS Carbon’s capabilities in evaluating materials under realistic conditions, as well as exploring other novel instrumentations pioneering new capabilities in this space, offering a glimpse into their potential impact on advancing carbon capture technologies. |
Case Study 1: Evaluating Quicklime for Direct Air Capture
| Direct Air Capture (DAC) represents a cutting-edge technology with the potential to play a pivotal role in combating climate change by actively removing carbon dioxide from the atmosphere. In this quest for scalable and efficient DAC solutions, the evaluation of materials like quicklime (calcium oxide, CaO) takes center stage. The detailed analysis of quicklime’s performance, as conducted through the DVS Carbon, offers critical insights into its suitability as a DAC sorbent. Understanding the kinetics and dependencies of quicklime’s CO2 capture in varying humidity levels and temperatures provides essential data for designing DAC systems that can operate under real-world atmospheric conditions. As the urgency to mitigate rising CO2 levels intensifies, the research into quicklime’s viability for DAC underscores the tangible impact of material science in the global effort to combat climate change |
Experimental Procedure:
Results & Insights:
Hydration and Carbonation Kinetics:The experiments revealed that the hydration and carbonation kinetics of CaO are highly influenced by the level of humidity. The higher the humidity, the faster the reactions, showcasing the material’s sensitivity to water content. This insight is critical for assessing the material’s performance in realistic flue gas conditions where varying humidity levels are encountered. Water Uptake and Thermal Regeneration:The recorded water isotherms provided valuable data on the material’s affinity for water. The subsequent thermal regeneration step demonstrated the feasibility of completely transforming CaO to calcium hydroxide, confirming the full extent of the process. Real-World Implications:Understanding the kinetics and dependencies on humidity allows researchers to tailor the use of CaO in direct air capture scenarios. The ability to control and optimize the carbonation process is crucial for the material’s practical application in industrial settings. |
Case Study 2: 13x Zeolite for Post-Combustion CO2 Capture
| The pursuit of effective post-combustion CO2 capture technologies is paramount in reducing greenhouse gas emissions from industrial processes and power generation. Within this landscape, the assessment of 13X zeolite’s performance, as explored through the DVS Carbon and BTA Frontier, bears significant real-world implications. This versatile material, known for its high CO2 uptake capacity, undergoes rigorous examination under varying conditions, including the presence of water vapor. Such research is pivotal for industrial applications, where flue gases often contain a mixture of CO2 and water vapor.
Understanding how 13X zeolite interacts with these gases informs the design of CO2 capture systems tailored for post-combustion settings, thereby contributing to more sustainable and environmentally friendly energy production. In essence, the study of 13X zeolite underscores the tangible contributions of materials science in the global endeavor to reduce carbon emissions and transition toward cleaner energy sources. |
Experimental Procedure:
Results & Insights:
Competitive Adsorption of CO2 and WaterSingle-Component Isotherms:The isotherms revealed that 13x exhibits a high uptake for both CO2 and water. However, even at low concentrations, water seems to saturate the material more rapidly than CO2. This highlights the competitive nature of water and its potential to impact the material’s performance in post-combustion scenarios. Multi-Component Breakthrough Experiments:The breakthrough experiments provided a dynamic perspective, showing that water and CO2 compete for adsorption sites. The total uptake of CO2 was significantly reduced when introduced simultaneously with water, emphasizing the need to consider water’s impact in practical flue gas conditions. Real-World Implications:These findings underscore the importance of understanding the water-CO2 competition when evaluating materials like 13x for post-combustion CO2 capture. The DVS Carbon and BTA Frontier tandem offers a holistic view, enabling researchers to gauge the material’s performance in complex industrial settings. |
The Breakthrough Analyzer (BTA) Frontier:
Illuminating Competitive Adsorption
| In tandem with the DVS Carbon, the Breakthrough Analyzer (BTA) Frontier stands as a pivotal instrument in the arsenal for comprehensively understanding the adsorption dynamics of materials, especially in scenarios involving competitive adsorption of multiple gases. |
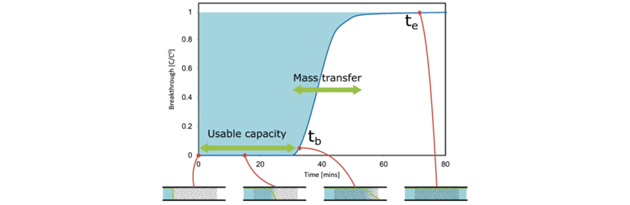
In a breakthrough analysis (BTA) method a known gas mixture is passed through a fixed bed or material, and the concentration is monitored at the end of a packed column of adsorbent. The shape of the resulting breakthrough curve provides equilibrium, bed transport and kinetic information. It is one of the few methods which can be used to determine true multicomponent adsorption data: e.g., individual uptakes of two or more components in a mixture.
Multi-Component Breakthrough Experiments:
Simultaneous Gas Introduction:Like the DVS Carbon, the BTA Frontier allows the introduction of multiple gases simultaneously, mimicking real-world conditions where various gases compete for adsorption sites. Dynamic Breakthrough Curves:By continuously monitoring the mass balance over a packed bed, researchers gain insights into how different gases interact with the material over time. |
Quantitative Uptake Analysis:
Integration of Breakthrough Curves:The breakthrough curves obtained can be integrated to quantitatively determine the total and individual uptakes of each gas. Realistic Industrial Conditions:The instrument enables experiments under conditions representative of industrial settings, providing actionable data for material evaluation. |
Versatility and Adaptability:
Customizable Experimental Parameters:Researchers can tailor experiments to specific industrial scenarios by adjusting parameters such as gas concentrations, flow rates, and temperature. Complementary to Gravimetric Analysis:When coupled with gravimetric instruments like the DVS Carbon, the BTA Frontier offers a holistic approach to understanding material behavior. |
Illuminating Material Performance:
| In the context of competitive adsorption, the BTA Frontier shines a light on how gases interact with a material in real-world scenarios. Its ability to simulate and quantify competitive adsorption provides invaluable insights for researchers and engineers aiming to deploy materials in practical applications, where multiple gases coexist and vie for adsorption.
Whether it’s elucidating the interplay between CO2 and water vapor in post-combustion flue gas or exploring the dynamics of other gas mixtures, the BTA Frontier adds a layer of depth to the understanding of material performance under complex industrial conditions. |

Conclusion:
The DVS Carbon emerges as a transformative instrument in the landscape of material evaluation for CCUS. Its ability to provide real-time, high-resolution data under diverse conditions empowers researchers to make informed decisions in selecting materials for carbon capture. As we navigate the complexities of decarbonization, tools like the DVS Carbon play a pivotal role in accelerating the development of sustainable solutions.
For a more in-depth understanding of the DVS Carbon and its applications, please download the instrument brochure below, or instead find out more on its produce page