Blog: Evaluating Materials for Carbon Capture, Unveiling the Power of DVS Carbon
The growing need to combat climate change has intensified the quest for sustainable solutions, leading researchers to explore innovative methods for carbon capture, utilization, and storage (CCUS).
Among the wide array of materials under scrutiny, solid sorbents have emerged as promising candidates, offering unique mechanisms for capturing and storing Carbon Dioxide (CO2). In this blog post, we unravel the transformative capabilities of the DVS Carbon, a high-resolution gravimetric sorption analyzer, and its pivotal role in evaluating materials for CCUS applications.

The Landscape of Solid Sorbents in CCUSAs the urgency to decarbonize our society accelerates, the scientific community is navigating a complex landscape of materials that have potential in addressing the challenges of carbon capture. From metal-organic frameworks (MOFs) to zeolites and activated carbons, researchers are exploring diverse avenues to find materials that can efficiently and sustainably capture CO2. The efficacy of these materials, however, hinges not only on their capacity to absorb CO2 but also on their performance under realistic and dynamic process conditions. Challenges in Material EvaluationEffectively evaluating materials for CCUS demands a multifaceted approach. Beyond assessing the quantity of CO2 a material can absorb, researchers must consider a spectrum of factors such as working capacity, kinetics, selectivity, stability, thermal properties, and cost considerations. A recent collaboration between researchers at Imperial College in London and industry experts resulted in the formulation of best practices, emphasizing the need for complete characterization metrics and in-process relevant conditions. |
The Crucial Role of DVS Carbon in Material Evaluation
In this landscape of complex challenges and evolving best practices, the DVS Carbon stands as stands out as a promising solution. Developed with the express purpose of enabling precise and realistic testing of materials for CCUS, the DVS Carbon is a gravimetric sorption analyzer that transcends traditional measurement tools. Let’s explore the key features that make the DVS Carbon an indispensable instrument in the pursuit of sustainable carbon capture solutions.
Gravimetric Precision and Real-Time MonitoringAt the heart of the DVS Carbon lies its gravimetric basis, leveraging a highly sensitive symmetric SMS Ultrabalance. This microbalance facilitates direct measurements of changes in sample mass with unparalleled precision, reaching down to the microgram level. This real-time monitoring capability with sub-second resolution is foundational in understanding the dynamic nature of material interactions with CO2 under varying conditions. Dynamic Flow System for Realistic ConditionsThe DVS Carbon’s dynamic flow system is a cornerstone of its capability to simulate realistic process conditions. By precisely controlling CO2 concentration, flow rates, temperature and humidity, researchers can mimic diverse scenarios, from Direct Air Capture with low CO2 concentrations to post-combustion conditions with high temperature and CO2 gas concentrations. This flexibility is instrumental in obtaining insights into material performance that align with real-world applications. Innovation in Sensor TechnologyTo enhance its performance, the DVS Carbon incorporates patented speed-of-sound sensors. This sensor technology, specially redesigned for this purpose, enables the high-accuracy measurement of carbon dioxide concentrations in the gas phase. Operating within a range of up to 100 percent CO2 by volume, these sensors contribute to the instrument’s ability to capture nuanced changes during absorption and desorption processes. In the following sections, we will delve into case studies that exemplify the DVS Carbon’s capabilities in evaluating materials under realistic conditions, as well as exploring other novel instrumentations pioneering new capabilities in this space, offering a glimpse into their potential impact on advancing carbon capture technologies. |
Case Study 1: Evaluating Quicklime for Direct Air Capture
| Direct Air Capture (DAC) represents a cutting-edge technology with the potential to play a pivotal role in combating climate change by actively removing carbon dioxide from the atmosphere. In this quest for scalable and efficient DAC solutions, the evaluation of materials like quicklime (calcium oxide, CaO) takes center stage. The detailed analysis of quicklime’s performance, as conducted through the DVS Carbon, offers critical insights into its suitability as a DAC sorbent. Understanding the kinetics and dependencies of quicklime’s CO2 capture in varying humidity levels and temperatures provides essential data for designing DAC systems that can operate under real-world atmospheric conditions. As the urgency to mitigate rising CO2 levels intensifies, the research into quicklime’s viability for DAC underscores the tangible impact of material science in the global effort to combat climate change |
Experimental Procedure:
Results & Insights:
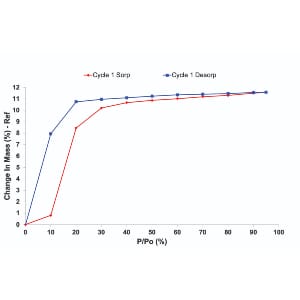
Hydration and Carbonation Kinetics:The experiments revealed that the hydration and carbonation kinetics of CaO are highly influenced by the level of humidity. The higher the humidity, the faster the reactions, showcasing the material’s sensitivity to water content. This insight is critical for assessing the material’s performance in realistic flue gas conditions where varying humidity levels are encountered. Water Uptake and Thermal Regeneration:The recorded water isotherms provided valuable data on the material’s affinity for water. The subsequent thermal regeneration step demonstrated the feasibility of completely transforming CaO to calcium hydroxide, confirming the full extent of the process. Real-World Implications:Understanding the kinetics and dependencies on humidity allows researchers to tailor the use of CaO in direct air capture scenarios. The ability to control and optimize the carbonation process is crucial for the material’s practical application in industrial settings. |
Case Study 2: 13x Zeolite for Post-Combustion CO2 Capture
| The pursuit of effective post-combustion CO2 capture technologies is paramount in reducing greenhouse gas emissions from industrial processes and power generation. Within this landscape, the assessment of 13X zeolite’s performance, as explored through the DVS Carbon and BTA Frontier, bears significant real-world implications. This versatile material, known for its high CO2 uptake capacity, undergoes rigorous examination under varying conditions, including the presence of water vapor. Such research is pivotal for industrial applications, where flue gases often contain a mixture of CO2 and water vapor.
Understanding how 13X zeolite interacts with these gases informs the design of CO2 capture systems tailored for post-combustion settings, thereby contributing to more sustainable and environmentally friendly energy production. In essence, the study of 13X zeolite underscores the tangible contributions of materials science in the global endeavor to reduce carbon emissions and transition toward cleaner energy sources. |
Experimental Procedure:
Results & Insights:
Competitive Adsorption of CO2 and WaterSingle-Component Isotherms:The isotherms revealed that 13x exhibits a high uptake for both CO2 and water. However, even at low concentrations, water seems to saturate the material more rapidly than CO2. This highlights the competitive nature of water and its potential to impact the material’s performance in post-combustion scenarios. Multi-Component Breakthrough Experiments:The breakthrough experiments provided a dynamic perspective, showing that water and CO2 compete for adsorption sites. The total uptake of CO2 was significantly reduced when introduced simultaneously with water, emphasizing the need to consider water’s impact in practical flue gas conditions. Real-World Implications:These findings underscore the importance of understanding the water-CO2 competition when evaluating materials like 13x for post-combustion CO2 capture. The DVS Carbon and BTA Frontier tandem offers a holistic view, enabling researchers to gauge the material’s performance in complex industrial settings. |
The Breakthrough Analyzer (BTA) Frontier:
Illuminating Competitive Adsorption
| In tandem with the DVS Carbon, the Breakthrough Analyzer (BTA) Frontier stands as a pivotal instrument in the arsenal for comprehensively understanding the adsorption dynamics of materials, especially in scenarios involving competitive adsorption of multiple gases. |
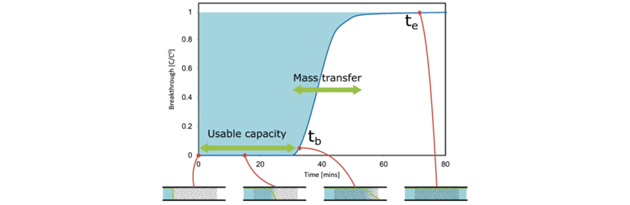
In a breakthrough analysis (BTA) method a known gas mixture is passed through a fixed bed or material, and the concentration is monitored at the end of a packed column of adsorbent. The shape of the resulting breakthrough curve provides equilibrium, bed transport and kinetic information. It is one of the few methods which can be used to determine true multicomponent adsorption data: e.g., individual uptakes of two or more components in a mixture.
Multi-Component Breakthrough Experiments:
Simultaneous Gas Introduction:Like the DVS Carbon, the BTA Frontier allows the introduction of multiple gases simultaneously, mimicking real-world conditions where various gases compete for adsorption sites. Dynamic Breakthrough Curves:By continuously monitoring the mass balance over a packed bed, researchers gain insights into how different gases interact with the material over time. |
Quantitative Uptake Analysis:
Integration of Breakthrough Curves:The breakthrough curves obtained can be integrated to quantitatively determine the total and individual uptakes of each gas. Realistic Industrial Conditions:The instrument enables experiments under conditions representative of industrial settings, providing actionable data for material evaluation. |
Versatility and Adaptability:
Customizable Experimental Parameters:Researchers can tailor experiments to specific industrial scenarios by adjusting parameters such as gas concentrations, flow rates, and temperature. Complementary to Gravimetric Analysis:When coupled with gravimetric instruments like the DVS Carbon, the BTA Frontier offers a holistic approach to understanding material behavior. |
Illuminating Material Performance:
| In the context of competitive adsorption, the BTA Frontier shines a light on how gases interact with a material in real-world scenarios. Its ability to simulate and quantify competitive adsorption provides invaluable insights for researchers and engineers aiming to deploy materials in practical applications, where multiple gases coexist and vie for adsorption.
Whether it’s elucidating the interplay between CO2 and water vapor in post-combustion flue gas or exploring the dynamics of other gas mixtures, the BTA Frontier adds a layer of depth to the understanding of material performance under complex industrial conditions. |

Conclusion:
The DVS Carbon emerges as a transformative instrument in the landscape of material evaluation for CCUS. Its ability to provide real-time, high-resolution data under diverse conditions empowers researchers to make informed decisions in selecting materials for carbon capture. As we navigate the complexities of decarbonization, tools like the DVS Carbon play a pivotal role in accelerating the development of sustainable solutions.
For a more in-depth understanding of the DVS Carbon and its applications, please download the instrument brochure below, or instead find out more on its produce page